The BMJ publication paves the way for FDA qualification of an AI tool
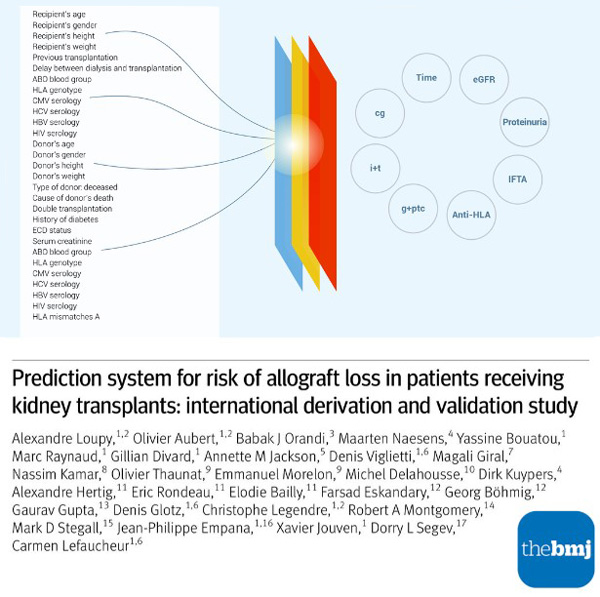
Developed by Professor Alexandre Loupy, nephrologist and Director of the Paris Transplant Group, the iBox system was trained and validated using data from 7,557 patients, ten European Union and the United States academic centres, and three randomised controlled trials. It predicts long-term allograft survival based on clinically relevant data, including kidney function, proteinuria, donor-specific antibodies, and biopsy results.
With European Medicines Agency (EMA) endorsement in 2022 and now FDA acceptance of its Qualification Plan in 2024, the iBox system is recognised globally as a reasonably likely surrogate efficacy endpoint for regulatory use in clinical trials. It has the potential to fast-track drug approvals, reduce reliance on long, costly trials, and transform care for kidney transplant patients.
- First transplant endpoint to receive FDA Biomarker Qualification Plan acceptance
- Built on The BMJ-published study using real-world data from 7,500+ patients
- Validated across EU and US centres and in three randomised controlled trials
- Enables five-year graft survival predictions at one year post-transplant
- Supports conditional drug approval and trial acceleration via the FDA’s Accelerated Approval Pathway
- Endorsed by both the European Medicines Agency (EMA) and FDA

This is a groundbreaking milestone—not just for transplant medicine, but for how AI can transform clinical research. It all began with a single BMJ publication, shaped by the journal’s exceptional editorial guidance.”
Professor Alexandre Loupy, MD, PhD
Director, Paris Transplant Group
Professor Loupy’s international derivation and validation study ranks in the top 5% of all research outputs scored by Altmetric, with coverage from 21 international news outlets and 263 citations in academic publications, according to Dimensions. This includes the KDIGO clinical guideline, Challenges in managing the kidney allograft: from decline to failure guideline published by Kidney Disease: Improving Global Outcomes (KDIGO), which has been shared through platforms like the KDIGO-ISN Webinar on Challenges in the Management of the Kidney Allograft, supporting global conversations on improving patient outcomes and providing essential guidance for nephrologists and transplant professionals.

“We desperately need new regulatory pathways that bring investment into transplantation. New therapies are essential to prolong the lives of transplant recipients and take the best care of the gift of life provided by our donors. This step is critical to enabling the dream of ‘One Kidney for Life’ to become a reality.”
Dr Karin Hehenberger
President of Lyfebulb patient engagement platform and a kidney and pancreas transplant recipient





